History of renal biopsy:
History of renal biopsy:
The kidney was one of the last full organs explored by percutaneous puncture-biopsy. For this deep organ, there was not only a risk of hemorrhagic complications of the kidney itself but also injury to another intra- or retroperitoneal organ (pancreas, colon, spleen, liver, suprarenal, large vessels, etc.). .). This injury could lead to the same hemorrhagic risks as the kidney but also other risks such as the perforation of a hollow organ.
The renal biopsy was thus long regarded as “inaccessible” and dangerous. Moreover, the revolutionary parallel discovery of the “artificial” kidney, treating renal failure, will hide for patients and doctors the interest of the diagnosis of kidney disease, limited then to autopsy pieces.
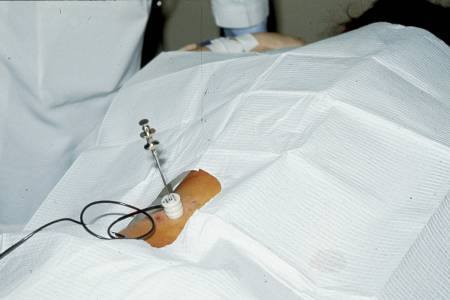
Percutaneous biopsy has been introduced efficiently since 1955 and has recently benefited from a considerable improvement in the identification of the kidney by imaging: it is the succession of various techniques using the abdomen without preparation, intravenous urography, isotopes, a shine enhancer and finally for ten years the ultrasound made either in the patient’s bed, or better in the ultrasound room.
Led by a radiologist under ultrasound, the technique has been improved. Since the law of 4 March 2002, it has become legally obligatory to carry out the biopsy with the “informed consent” of the patient signing the agreement and using expert hands to collect the sample under ultrasound control.
Means of study of the renal biopsy – History:
The introduction of different study techniques has considerably increased the richness of the histological interpretation of the biopsy.
The two techniques of optical microscopy and electron microscopy were introduced almost simultaneously from 1956. This technique allowed the analysis of the normal human kidney and the glomerular morphological lesions.
It has mainly served experimental protocols.
In Europe, several teams contributed to its development.
It is in Europe of P. Iversen in Denmark, R.-H. Hepstinstall in London, J. Hamburger, R. Habib, J. Berger and N. Hinglais in Necker, J. Bariety in Broussais and L. Morel Maroger in Tenon, J.-M. Suc in Toulouse and J. Traeger in Lyon. They all contributed to the description of the various nephropathies and their treatment.
The qualitative leap is made 10 years later in 1966 by the routine introduction of immunohistochemistry requiring a frozen sample used either for labeling methods using radioactive isotopes or for enzymes such as peroxidase, immunofluorescence. This stage, begun by Coons in 1941, brings nephrology into a new era, that of the immunological marking of the kidney.
It was the longest and most difficult stage to develop, spanning about 20 years. A number of big names must be cited as Coons et al. in 1941, Hill et al. in 1953 and 10 years later, Nairn et al. All understood that many glomerulonephritis responded to antigen-antibody conflict and invented immunohistochemical tagging techniques introducing the first antibodies. These first antibodies were then synthesized in the research laboratories of hospitals recognizing a kidney antigen. Another difficulty was to invent a microscope suitable for the screening of certain fluorescent substances marking the antibody, making it “visible” to the eye by using an ultraviolet lamp, connected to the microscope.
The substances in question may be fluorescent green if the isocyanate or fluorescein isothiocyanate is used, or in red with rhodamine-lissamine B. The first applications were intended to detect microbial, viral, parasitic and fungal antigens as well as circulating antibodies.
The succession of this brilliant but artisanal approach was made by the industry around the 1970s, and allowed an explosion of means of investigation from commercial antibodies “fluorescent” or marked by peroxidase. These antibodies have become of better quality, making it possible to increase the sensitivity and the specificity of the marking.
Later, interest was focused on the normal components of the kidney such as renin and also on the nature of the glomerular deposits called “immune complexes” consisting of immunoglobulins (Ig) and complement fractions, their location and morphological appearance.
Finally, in the 1980s morphometry and confocal microscopy improved the techniques of measurement and marking.More recently and still in development, molecular biology techniques will allow to study the diversity of the genome and its expression from the renal biopsies.
Place of renal biopsy in the diagnosis and prognosis of renal disease:
Renal biopsy is the key to the histological diagnosis of glomerular and tubulo-interstitial diseases. It can therefore have therapeutic consequences. Glomerular involvement is the most frequent lesion, difficult to assess, and is either a primary renal disease or a disease within the scope of systemic diseases. Because of their vascular nature, glomeruli are capillaries forming an arterial portal system located between the afferent arteriole and the efferent arteriole of the glomerulus through which almost all renal blood flow passes, ie 20% of the cardiac output. By its filter function between the blood and urinary space, the glomerulus is continuously exposed to plasma proteins. The glomerulus behaves not only as a passive filter but as a privileged site where circulating molecules such as immune complexes can be deposited.
The identification of glomerular deposits and certain antigens has made it possible to address the pathogenesis of glomerulopathies, to propose a classification of glomerular diseases and to distinguish between inherited glomerulonephritis and glomerulonephritis that can be treated. Some glomerular lesions are classified according to international consensus such as lesions of lupus or lesions of systemic vasculitis. For the classification of lupus glomerulonephritis under the auspices of the World Health Organization (WHO), the first classification dates back to 1957 and the last was in 2004 with the participation of the International Society of Nephrology and the Renal Pathology Society ISN / RPS 2004).
For systemic vasculitis, this is the classification of Chapel Hill.
Sampling techniques and means of routine study of renal biopsy:
Histological sampling always requires two different samples for the techniques of optical microscopy and immunohistochemistry (immunofluorescence). In cases where electron microscopy is indispensable, it will be possible to carry out a third sampling, or the ends of one of the fragments may be cut. All these samples will be processed in different packaging methods.
Optical microscopy:
Attachment of the biopsy sample:
The renal biopsy has benefited in Europe and especially in France from a particular fixative different from the formalin used in Anglo-Saxon countries including the United States. Formalin is a universal fixative of all biopsies and surgical specimens.
The most commonly used fixative in Europe until recently was the Bouin alcoholic liquid or the Dubosq-Brazil liquid which allows certain colorations useful for diagnosis. Currently it is replaced in France by a fixative associating formaldehyde with alcohol and acetic acid (alcohol-formaldehyde acetate: AFA). The utility of these fixatives is to allow Masson trichrome, which is the color of excellence, to demonstrate immune deposits, a feature of the majority of glomerulonephritis. The immersion fixation in the AFA is immediate and continues for 2 to 3 hours. Beyond this period, the fixer is replaced by 70% alcohol. One of the reasons for the Anglo-Saxon preference for formalin is the destruction by Dubosq-Brazil of epitopes “accessible” to marking.
This prevents certain immunolabelings necessary for diagnosis and especially for research. Eosinophil granulations also disappeared with Dubosq-Brazil.
Inclusion is currently practiced in PLCs. The inclusion medium is a mixture of Paraplast® and paraffin. The inclusion is carried out after sampling in four liquor baths (15 minutes each) and three baths of toluene or xylene (20 minutes each).
Serial and numerous sections are made with a microtome, with a thickness of 2 to 5 μm. The ribbon is collected in its entirety so as not to risk “missing” focal lesions. All cuts are recovered by depositing five per blade, keeping material for possible future techniques.
Cuts of the cuts:
Before discussing the histological interpretation, we give the definition of “immune deposit”. The “immune” deposition is defined in histology as an extracellular deposit, brick red with Masson trichrome, homogeneous and osmiophilic, containing in immunofluorescence Ig and fractions of the complement. In electron microscopy, it appears to be “dense” to electrons, osmiophilic and non-argyrophilic. This definition therefore involves all three techniques. In common practice, the first two are sufficient for diagnosis, justifying the abandonment of systematic sampling for electron microscopy.
Masson’s green (or blue) trichrome, Schiff periodic acid and periodic acid (PAS) are the three colorations used for diagnosis. The trichrome shows the immature deposits in red brick (Mallory red), the basal membranes and the mesangium in green (light green), and in violet the nuclei (hematoxylin). The silver colourations, according to the techniques of Jones or Marinozzi, color black-brown matrices such as glomerular basal membranes (MBG), tubular as well as the mesangial matrix. Silvering confirms the endocapillary cell proliferation with its appearance in “cubicles” of the mesangial areas.
It shows the modifications of the walls with a “double contour” appearance in the form of two lines. For extramembraneous deposits, it is the matrix material surrounding the deposit that gives these aspects of spicules, clubs or craters or chains. In the Alport syndrome, defects of silvering are also detectable. Hematoxylin-eosinesafran is a basic stain that analyzes the resident cells or infiltrates the glomerulus. The PAS pastes the mesangial matrix in pink, the MBGs and the brush edges of the tubes.
It makes it possible to distinguish so-called “hyaline” deposits from fibrinoid deposits. Staining by the May-Grünwald-Giemsa can help with cell analysis. Many specific colorations can be added. This is, for example, that of amylose.
It can be Congo Red, Violet Crystal or Thioflavin.
The crystals may require additional reading in polarized light which visualizes the structure and the specific pattern of the crystals or a complementary study in crystallography on thick sections. The coloration of the fats (red in oil, black Sudan) is done on the frozen sample.
Histological lesions:
The histological lesions sought within a glomerulus are either focal, affecting less than 50% of the glomeruli analyzed, or diffuse affecting more than 50% of the glomeruli. Within a glomerulus, the lesion is segmental, if only part of the glomerulus is affected, or global if the entire glomerulus is affected. The term endocapillar proliferation corresponds to a hypercellularity of the flocculus in which the endothelial cells, the mesangial cells and sometimes the circulating inflammatory cells (polymorphonuclear cells, mononuclear cells) participate. The MBGs have “double contour” aspects with accumulation of endomembranous deposits also present in the mesangium and / or extramembrane deposits located on the external side of the wall. These “double edges” stained by silvering correspond to the slippage of the mesangial cell between MBG and endothelial cells.
Finally, foci of fibrinoid necrosis exist that can be associated with lesions of the flocculus or with croissants corresponding to an extracapillary proliferation filling the urinary chamber. These lesions are caused, among other things, by the proliferation of cells in the Bowman capsule. They are called focal necrotizing glomerulonephritis with pure extracapillary crescents or glomerulonephritis and are often encountered in systemic vasculitis. Inflammatory and fibrous lesions are of interest to the glomeruli, the tubes and the interstitium. Arteries, tubes and interstitium are also the site of specific lesions.
Glomerular immune “deposition” may be present in many endomembranous sites: mesangial, intraparietal, intraluminal with fibrin thrombi or immunoglobulin plugs obliterating capillary lumens. It may also be in an extramembrane and / or hump situation on the external side of the wall. The tubes are also the site of a “dense” chromophilic deposit of C3, identical localization during a deposit of a monoclonal Ig, in the interstitium and the vessels. Spotted in optical microscopy, they share their “extracellular” situation with immune deposits and can be easily identified by freezing.
Immunofluorescence on frozen material:
The immune origin of a glomerulopathy is currently accepted on the sole finding of the deposit of Ig coded with certain fractions of the complement, even if the antigen is unknown and not detected. By analogy with the acute serum experimental disease, in which circulating immune complexes appear and persist, the latter have long played a part in the genesis of glomerulonephritis in man. It may be a passive deposition of immune complexes circulating through the glomerular filter or a more specific deposition involving certain characteristics of the complexes such as size, greed or load. Currently, it is preferable to evoke another mechanism: that of immune complexes formed in situ. Immune deposition would be secondary to the in situ binding of a free antibody circulating on a glomerular antigen. The glomerular antigen may be a structural antigen or a molecule that is attached for non-immune reasons to a glomerulus structure. This is referred to as a “planted antigen”. For example, a cationic antigen may bind in the anionic MBG or a lectin such as concanavalin A on the sugars of this wall.
Freezing:
The fragment can be frozen by rapid immersion in isopentane cooled by liquid nitrogen or directly in liquid nitrogen.For some it is placed in Tissutek, which hardens on contact with liquid nitrogen. When the sample is to be transported to another laboratory, it is transported in a plastic tube to the cold with dry ice and is frozen on site, avoiding thawing.
“Direct” immunofluorescence of frozen tissue:
It is a rapid technique that requires only 2 hours, which is valuable in certain pathologies requiring urgent results and treatment such as rapidly progressive glomerulonephritis. The sections made in series of the fabric with a thickness of 2 to 3 μm are obtained with a cryostat. An “incubation” of the antibodies on the sections (unattached or fixed to acetone for 10 minutes) takes place in a humid atmosphere for 30 minutes and then the preparations are rinsed with a buffer. The assembly is made with buffered glycerine. The sections are examined with a microscope equipped with an ultraviolet lamp. Perishable preparations are photographed immediately. The “green vivid” fluorescence extinguishes under the rays in less than 2 minutes, requiring to look at them quickly and to keep them away from light and cold. On the other hand, the remainder of the frozen sample is kept in the liquid nitrogen for years, and can be reused if necessary for further investigations under the same conditions.
Results and routine interpretation of direct immunofluorescence:
Immunological marking of “immune” intrarenal deposits in immunofluorescence:
For the routine and systematically, at least eight fluorescent polyclonal commercial antibodies are used. These antibodies are directed against the immunoglobulin heavy chains, against the two lambda and kappa light chains, against certain complement fractions such as C3, C1q, C4, fibrinogen and for a number of laboratories, an antibody directed against the anti – albumin. Other antibodies may be needed routinely, for example for acute “humoral” rejection in a transplant, anti-C4d is added to the other antibodies. The constituents of MBG can be studied. Abnormal distribution of the alpha chains of collagen IV makes it possible to diagnose Alport syndrome.
In a resounding editorial, Dixon in 1968 imposed a pathogenic classification. Even criticized because it forgets cellular immunity and gives too much room for humoral immunity and immune complexes often absent, it is still used routinely.The three major pathogenic reference categories are still in use:
• the linear labeling of MBGs, which must make it possible to look for circulating anti-MBG antibodies;
• “granular” deposits of glomerular immune complexes in immunofluorescence: taking into account the immunochemical nature or composition, size, topography on one side or the other of the MBG or in the mesangium and the grouping of the different deposits;
• predominant granular mesangial deposits of IgA.
The presence of complement in the kidney is in favor of the deleterious immunological conflict for the glomerulus, in particular the C5-b9 terminal complex. Complement may be activated by the classical pathway involving immune complexes such as lupus, type I glomerulonephritis (GNMP), cryoglobulinemia (where C3 and C4 are low). The alternate pathway is activated by factors other than immune complexes in Berger’s disease, type II GNMP, and certain post-infectious glomerulopathies. There is cellular recruitment of inflammatory cells, particularly polynuclear and monocyte macrophages within the glomerulus, close to the deposits. The phenotype of inflammatory cells and other interstitial cells is studied on a fixed material by revealing diaminobenzidine (DAB) using an automaton, with amplification if necessary, with the antibody battery of the laboratory of general pathology.
For a number of glomerulonephritis, the antigen has been identified. This is the case for certain extramembraneous glomerulonephritis, GNMP, bacterial and viral infectious glomerulonephritis, and some glomerulonephritis associated with cryoglobulinemia types I and II, which are detailed below.
How to interpret immunofluorescence routinely?
There are immunofluorescence markings with only one diagnosis:
Absence of marking:
It eliminates deposited glomerulonephritis. The presence of very fine mesangial filaments of IgM associated with a nephrotic syndrome leads to the diagnosis of idiopathic “minimal glomerular lesions” (LGM) in children. The MBG are finely highlighted by albumin. The LGM is often corticosensitive. In the case of corticoresistance, the molecules characterizing the podocytes can be studied (podocine, nephrine, dystroglycans, a 3 b 1 integrin …). The etiology in adults requires to eliminate its association with certain neoplasias including Hodgkin’s disease. If there are episodes of recurrent macroscopic hematuria, renal insufficiency, hypoacousia, the alpha chain test of collagen IV should be done.
Linear fixation of glomerular basal membranes in immunofluorescence:
The classic form of Goodpasture’s syndrome is hemorrhagic glomerulopathy associated with pulmonary haemorrhage associated with type IV anti-collagen autoantibodies directed against MBG. The antigen is located in the alpha-3 chain of collagen IV to the non-collagenic domain (NCI). Immunofluorescence shows a linear attachment along the MBG, sometimes associated with linear labeling of the tubular glasses of the distal tubes with anti-IgG antibodies, both light chains and anti-C3. There is also a marking of the urinary chamber and the extracapillary proliferation with the antifibrinogen antibody.
Necrotic glomerulonephritis with extracapillary proliferation progresses rapidly towards kidney destruction. Treatment initiated in an emergency is aimed at eliminating the circulating toxic antibodies.
Rare and of different evolution, there are glomerulonephritis associated with the same linear labeling in immunofluorescence, but involving other anti-MBG antibodies directed against other domains of collagen IV. The specificity of the epitope recognized by the antibodies is important for the scalability of the disease which is less severe with the other anti-MBG antibodies.
Diagnosis of nephropathy with IgA deposits:
It is done by immunofluorescence. It shows constantly granular deposits drawing the mesangial areas in all glomeruli associated with the other Ig and C3. This aspect is typical of Berger’s disease initially described by Berger and Hinglais in 1968. It remains the most common chronic glomerulonephritis in France and the most widespread in the world. Mesangial immunofluorescence is identical regardless of the histological form, which it shares with rheumatoid purpura. This is different for large proliferative forms where marking becomes more diffuse in the mesangioparietal situation. Other etiologies of mesangial IgA are chronic hepatopathies especially alcoholic and / or certain inflammatory intestinal diseases.
Granular deposits of glomerular immune complexes:
Extramembraneous glomerulonephritis:
It is defined by the presence of constant small granular deposits of IgG and C3, situated on the external side of the MBG under the podocytes (thus pure “parietal”).
The extramembrane glomerulonephritis stage I shows a pseudolinear aspect of IgG. It represents 20% of adult chronic glomerulonephritis and is most often idiopathic. The list of secondary forms is long with mainly systemic diseases like lupus, mixed connectivites, sarcoidosis and rheumatoid arthritis.
In the latter case, it was possible to show the responsibility for the treatments used (gold salts, D-penicillamine) for its appearance and for its regression when the toxicant stopped. Some thyroids are present in the thyroglobulin deposit.For infectious extramembranous glomerulonephritis, hepatitis B virus, or treponemal disease was found in syphilis.During paraneoplastic extramembraneous glomerulonephritis, where carcinomas dominate, rare tumor antigens have been eluted.
For lupus, it represents in its “pure” form the class V of the last classification of the ISN / RPS 2004 of the glomerulonephritis. The diagnosis of lupus extramembranous glomerulonephritis is referred to the presence of C1q in glomerular deposits, and the lesser marking often present of a tubular basal membrane or a vessel or of the mesangium with anti-IgG.
Antigen intervention in experimental models and in some human forms of extramembrane glomerulonephritis could be determined. In this context, the renal biopsy allowed to approach the pathogenic mechanisms of these human forms of extramembraneous glomerulonephritis.
For example, Ronco et al. have shown, in the extramembraneous glomerulonephritis of Heymann nephritis in the rat, that the antigenic target is a structural antigen called megalin. The antigen is expressed both by the tubular cells of the proximal bypass tube and at the surface of the podocytes, where the in situ formation of the immune deposit takes place. The same group recently identified the first human antigen, a neutral endopeptidase also from the brush border, involved in a prenatal form of human extramembrane glomerulonephritis, from alloimmunization in a mother deficient in this enzyme.
Post-infectious glomerulopathies with “humps”
Described mainly in children or young adolescents, acute poststreptococcal glomerulonephritis shows a “starry” appearance of C3 and / or IgG in parietal and mesangial situations with bulky humps in balls or cones on the external side of the MBG. It has analogies with experimental serum sickness. The antigen belongs here to certain nephritogenic streptococci present in the immune complexes. Evolution is often reversible in these large endocapillary exudative forms with polymorphonuclears in the glomerular capillaries.
In addition to skin and otolaryngological infections in the healthy child, the portals and germs have multiplied with so many different clinical aspects. These may include deep suppurations difficult to highlight or specific entry gates such as endocarditis or localization to a shunt. They can occur on fragile lands in intensive care units or in alcoholics or drug addicts using heroin. All these factors modify the type of glomerulopathy sometimes segmental and focal but still very rich in deposits of C3.
Very recently, a new aspect of immunofluorescence has been described in diabetic patients with nodular glomerulosclerosis during staphylococcal infection. In this case, what dominates is a “starry sky” marking of IgA superimposed on C3.
Glomerulonephritis membranoproliferative:
It is defined by mesangial and parietal endomembranous deposits (along the MBG), a complement associated variable with IgG, IgA and IgM and with a hypocomplement.
Two main types are observed. Type I GNMP and Type II GNMP in their classical form are lobular. In immunofluorescence, extramembranous deposits, sometimes hump, may be found, which are rather irregular. It is currently considered that the diagnosis of type I GNMP should seek cryoglobulinemia which sometimes assumes a characteristic morphology. Then, the mesangial proliferation and the “double contours” of the Ig and C3 plugs are added to the capillary lumen. This makes it possible to identify within the glomeruli the deposited type II cryoglobulinemia containing IgG, IgM and C3. In 1989, hepatitis C virus (HCV) was identified by Choo et al. These GNMPs were then reported to the renal deposition of cryoglobulinemia II related to HCV infection. The viral ribonucleic acid (RNA) is concentrated within cryoglobulin in plasma and in renal cryoprecipitate. It is a model of deposition of circulating immune complexes with activation of the classical complement pathway. The rheumatoid factor, which is a monoclonal IgM, induces rapid aggregation of immune complexes that are not eliminated but aggregated and precipitate in capillaries including glomeruli. The work of Izui et al. showed that deposition in the kidney and skin depended on two properties of cryoglobulin: anti-IgG activity related to the rheumatoid factor, and the quality of ability to precipitate in the cold.
Another very rare GNMP (GNMP type II), described by Galle and Berger in 1962, is characterized by a morphological marker, the “dense deposition” or intramembranous deposit, in all kidney basals. This deposit presents a characteristic aspect in optical and electronic microscopy. In the serum, an autoantibody, the C3 Nef, permits the permanent activation of the alternate pathway. Immunofluorescence shows characteristic C3 marking by a rail aspect of the walls and an intense fixation of C3 into mesangial balls and a granular labeling of C3 on the membranes of the tubes.
Another type of GNMP with isolated deposits of C3 without dense deposition was individualized with a particular aspect in optics and immunofluorescence. This aspect shows a granular marking of mesangial and parietal C3, tubular glasses and Bowman capsules corresponding to this so-called “granite” material which remains of unknown composition.
Systemic diseases of different immunological mechanisms:
With “immune complex” (systemic lupus erythematosus [LED] and rheumatoid purpura):
LED. It presents as a general disease with several visceral localizations and a renal involvement in 50 to 70% of the cases. The biopsy shows diffuse deposits with the presence of IgG, IgA, C3 and C1q in all renal structures. This type of glomerular and extraglomerular marking evokes only one diagnosis, that of lupus. Identical deposits are observed in other organs affected by the disease such as the choroid plexus, heart, lungs, skin, etc. In glomeruli, these may be large endomembranous deposits in plugs filled with capillary lumen or wire-loop thickening walls or parietal grains of extramembrane deposits.
They can draw the mesangium and the Bowman capsules. All the architecture of the renal parenchyma is drawn by the fluorescent IgG. Apart from the glomeruli, the deposits are granular, and underline the tubular basals, marking the interstitium in the form of grains, small clusters in the media of the vessels. They are sometimes also linear along the peritubular capillaries.
Most often, there is a lupus glomerulopathy where the marking is only glomerular almost always associating the same Ig including IgG and C1q. The classification of the ISN / RPS 2004 of the glomerulopathies of the LED does not take into account the results of the immunofluorescence.
It is interesting to note another viral glomerulopathy called “pseudolupic”. It is an affection rich in IgG with sometimes an extraglomerular marking. It has been described in the first patients infected with the human immunodeficiency virus (HIV) untreated and even more in the drug addicts co-infected with the HCV.
Rheumatoid purpura. It is a systemic vasculitis which associates with renal involvement (nephropathy with deposits of IgA) a cutaneous vascular purpura with angéite with deposits of IgA and arthralgias. It is also associated with digestive impairment, more rarely pulmonary or neurological. Biopsies from various visceral sites contain IgA deposits in the vessels.
Other systemic diseases without deposition of immune complexes:
Necrotizing glomerulonephritis with crescents (or pure extracapillaries) “pauci-immunes”. They are encountered in the context of systemic vasculitis such as microscopic polyangiitis, Wegener’s disease, or more rare Churg and Strauss disease. These lesions can also be isolated. The classification of systemic vasculitis is based on the size of the affected vessels. The Chapel Hill classification is currently used. In 1985, in this group, autoantibodies against molecules of the cytoplasm of neutrophils or ANCA were detected in 85% of patients. ANCA are detected by indirect immunofluorescence on polynuclear smears. Two types are recognized: the perinuclear type (p-ANCA) is directed against the myeloperoxidase; the cytoplasmic type (c-ANCA) is directed against proteinase 3. The role of these ANCAs in the pathogenesis of vasculitis is not fully understood but can be used as markers of disease activity. The extrarenal signs are arthralgia and fever. It may also be other visceral sites such as the upper airways and the lung.
These localizations also play a role in prognosis. In immunofluorescence, they are poor in immunoglobulins and in C3, which is often very focal. On the other hand, fibrin marks the growths or extracapillary proliferation that can fill the entire urinary chamber of the injured glomeruli and eliminates Goodpasture syndrome due to the lack of linear anti-IgG binding along the MBG. The histology makes it possible to envisage the prognosis by locating the number of normal glomeruli. In the kidney, glomerular involvement may be associated with necrotizing angiitis of arterial or interlobular arteries. This is a therapeutic emergency, the results of which are followed by a European group.
Hemolytic uremic syndrome (HUS).
The appearance in immunofluorescence is unique, supplementing the lesions of microthromboses of glomeruli, arterioles and small arteries which are diagnosed on optical microscopy. There are immunofluorescence deposits of fibrin in the mesangium and along the capillary walls of the injured glomeruli. They are also present in the vessel wall and in the thromboses. These are readily C3, IgM, and von Willebrand factor.
Segmental and focal hyalinosis (HSF) lesions of glomeruli in adults. In immunofluorescence, the primary or secondary or familial HSF lesion shows irregular marking of the glomerular segmental lesion by IgM, C3 and C1q. Visceral epithelial cells have generally lost their podocyte phenotype.
Observed in the course of HIV with a particular genetic susceptibility of certain black ethnic groups, the glomerulopathy with “collapse” shows in immunofluorescence either a marking of the lesions constituted of HSF, or very little in the recent forms. In this case, it may be IgA in many hyalinized podocytes undergoing necrosis and detachment.
Diabetes. It has specific labeling with polyclonal IgG and albumin. The marking is linear along glomerular and tubular basalts and mesangial nodules. The study in immunofluorescence is fundamental because the appearance in light microscopy can be confused with lobular glomerulonephritis and nodular glomerulosclerosis with deposits of light chains and / or heavy chains of immunoglobulins.
Amylose non AL. AA amyloidosis is, after AL amyloidosis, relatively frequent in the kidney. A diagnosis of glomerular amyloidosis requires its obligatory typing to guide the etiologic research of the clinician. Any amylose will be studied systematically (the two light chains have been studied in immunofluorescence) with the following antibodies: anti-SAA, antitransthyretin. According to clinical orientation according to ethnic origin and / or family forms, tissue and blood samples are referred to specialized biochemistry and genetics laboratories.
Immunological marking of “non-immune” intrarenal deposits:
Renal biopsy and immunofluorescence during dysproteinemias and / or organically deposited glomerulonephritis (GOMMID):
Glomeruli are the preferred target of the deposition of circulating proteins produced during systemic malignant hematological diseases (called dysproteinemias).
They are associated with B lymphocyte proliferation. Most often, it is myeloma, Waldenström disease, certain lymphomas (chronic lymphocytic leukemia, marginal zone lymphoma, etc.).
In some cases, no serum dysglobulinemia is found.
The nephropathies with deposits of monoclonal Ig are multiple with:
• myeloma tubulopathy with light chain cylinders;
• Fanconi syndrome with Ig crystals in the proximal convoluted tubules;
• AL amyloidosis;
• deposition of light chains and / or heavy chains along basal membranes of tubes, glomeruli and kidney vessels (Randall’s syndrome);
• glomerulonephritis of monoclonal cryoglobulinemia type I (more often IgG than IgM);
• and also GOMMID.
All these Ig deposits are identified on morphological characteristics, including specific staining and the results of renal immunofluorescence on freezing with study of heavy and light chains of Ig. Serum peak analysis is also associated with immunoelectrophoresis, the search for circulating cryoglobulinemia, light chains in the urine, the study of medullary and circulating lymphoplasmocytic populations, and the molecular analysis of Ig chains.
Immunofluorescence is essential for diagnosis: marking of a myelomatous cylinder, isolated tubular reabsorption (with or without a cylinder) of a single light chain, or marking of glomerular or vascular AL amyloidosis with the lambda chain more frequently than kappa, which is on the other hand more frequent marking tubular and glomerular basal in a light chain deposition disease (LCDD). The same is true for the components of cryoglobulin type I in immunofluorescence, which is also accompanied by optical microscopy of a particular GNMP, even specific. It is also interesting to study the isotype (eg IgG) in the glomerular deposition in immunofluorescence with the different subclasses of the gamma chain showing, in the rare cases of the literature where the subclass of anti- IgG was studied, most often (this is also our experience) of an IgG 3 kappa (more often than an IgG 1 kappa), always associated with C3 in the same territory.
In fact, a new lesion appears as evocative of this GOMMID, realizing in optical microscopy a particular form of GNMP called “atypical” due to the presence of numerous and extramembraneous deposits (silvering), with immunofluorescence of large “parietal” of the deposited monotypic material. These are usually IgG 3 kappa associated with C3. Uzui et al. have developed a murine model of monoclonal cryoglobulinemia type I with systemic vasculitis and a typical glomerulopathy of cryoglobulinemia, very similar to human lesions, from hybridomas producing monoclonal antibodies of isotype IgG 3 .
The ultrastructure shows specific lesions in these pathologies with monoclonal Ig deposits either in the form of amylose fibrils or in the form of very dense osmiophilic granules during the Randall syndrome. They may also be “organized” deposits observed during the “immunotactoid” glomerulonephritis describing the crystalline organization of these Ig deposits. The term GOMMID is preferred to that of immunotactoids, with in ultrastructure deposits consisting of rectilinear microtubules with a visible central lumen. In the absence of cryoglobulin and / or other detectable circulating protein, electron microscopy identifies these deposits of type I cryoglobulin or deposits belonging to a GOMMID.
Renal transplantation, humoral rejection and C4d intake:
For the past ten years, humoral rejection, initially unknown and ignored, has been better diagnosed. Its incidence during the acute rejection of graft is of the order of 20 to 30%. It is linked to the presence of circulating anti-human leukocyte antigen (HLA) antibodies directed against the donor (donor-specific antibodies [DSA]). Its frequency is increasing after renal transplantation, parallel to the risk factors that are polytransfusions, pregnancies or abortions and previous transplants. In parallel with the considerable improvement in immunological techniques for the identification of antibodies and their specificity in blood, the discovery of C4d as a marker of humoral rejection occurs.It is the specific and exclusive marker of solid organ rejection by anti-HLA anti-donor antibodies. The search for C4d is done routinely on frozen material, and more rarely on fixed material. The histological lesions of the humoral rejection, its correlations with the presence of anti-HLA antibodies specific to the donor and the presence of C4d have been reported from 2002 and allowed the update of the Banff classification reviewed every 2 years.
Electron microscopy and routine human renal biopsy:
In the late 1950s and 1960s, the systematic joint practice of traditional optical microscopy, immunohistochemistry (immunofluorescence), and transmission electron microscopy (TEM) resulted in the definition of the biopsied human kidney initial lesions of nephropathies and to classify them. More than a century of cadaveric pathological anatomy had not made it possible.
Acquisition of transmission electron microscopy:
MET learned to read and interpret precisely the results provided by optical microscopy and immunofluorescence.
It imposed almost permanently the histology of the renal parenchyma, particularly the glomeruli. Many elements have been proved. It is the existence of the mesangium long ignored or disputed, the fenestrated character of the endothelial cells, the shape of the podocytes with a body floating in the urinary space extended by feet and pedicels inserted on the external part of the MBG limiting epithelial slits. The existence of slit diaphragms stretched between neighboring pedicels along MBG, the structure and dimensions of the MBG and the mesangial matrix and the ultrastructure of the juxtaglomerular apparatus have also been defined.
MET demonstrated the structure and location of pathological deposits. These may be homogeneous or finely granular and called immune (electron-dense, osmiophilic, non-argyrophilic deposits) or fibrillar, such as the amyloid substance formed of fine fibrils deposited without orientation or the immunotactic deposits formed of parallel microtubules. Their localization is defined: extramembrane, endomembranous or mesangial. Multiple cell alterations have been described.The spread of pedicels with the disappearance of numerous epithelial slits and slit diaphragms, the proliferation and interposition of mesangial cells between MBG and the endothelial cell explaining the “double contour” images of the walls of the glomerular capillaries observed in MPGN. The MET also analyzed the structure of pathological MBGs in certain congenital and hereditary diseases (Alport syndrome), their thickening (glomerulosclerosis) or conversely their thinning.
Constraints:
The establishment and operation of an electron microscopy laboratory is expensive. This laboratory must include a dark room for the electron microscope, ventilated or air-conditioned, stable and protected from vibrations (attention to the proximity of elevators!), An obscure room for photographic development. A room must be reserved for the preparation and must have many facilities and materials like an ultramicrotome with diamond knives, an optical microscope, a precision balance, a pH meter, a centrifuge, graduated ovens, a refrigerator and a hood with evacuation system. The practice of electron microscopy is only possible if it is entrusted to qualified persons and to a laboratory that is trained in sample preparation and reading.
The preparation of a human renal sample by MET has unavoidable constraints. In addition to the fragments reserved for optical microscopy and immunofluorescence, it requires the production of a third fragment which is technically specialized. Unlike in animals, human biopsy specimens can not benefit from optimal fixation techniques such as in situ fixation or controlled fixation by renal arterial perfusion. This is the reason why ultrastructural images obtained in animals are often much better than those obtained from human samples taken by immersion.
It is not surprising that on human kidney biopsies the glomerular capillary loops are not regularly rounded with little or no red blood cells or if the lumen of the proximal contoured tubes is collapsed or not very open.
This is due to a compulsory artefact in the biopsy specimens even quickly fixed by immersion. Upon removal, the renal tissue contracts and expels some of the blood and urine it contains. In addition, the fixatives indicated for electron microscopy penetrate hardly into the parenchyma. It must therefore be fragmented sufficiently with the razor, which increases the risk of artefacts.
The following preparation technique is one of the most common. In order to be properly fixed, fragments 0.5 to 1 mm apart are immediately immersed in glutaraldehyde prepared extemporaneously at 2.5% in a cacodylate buffer or phosphate buffer. This fixation is followed by fixation with 2.5% osmium tetroxide in the same buffer. The fragments are dried in ethanol at increasing concentrations and then in propylene oxide. The fragments are included in gelatin capsules containing an Epon resin. Epon is polymerized in an oven at 60 ° C. for 48 hours. 1 μm “semifine” sections stained with toluidine blue are used to select areas of interest on which ultrafine sections are deposited on metal grids.
These sections are stained before reading with uranyl acetate or lead salts. Other colorations are sometimes necessary: by phosphotungstic acid or by the silver salts.
This long and meticulous technique shows that in electron microscopy one can analyze what one is looking for but it is difficult to envisage a reading of the samples available in extenso. The analysis of four glomeruli by biopsy requires considerable reading time.
Indications of MET in 2007:
MET is an indispensable research and investigation method, particularly when associated with scanning electron microscopy or when using ultrastructural immunohistochemistry techniques with antibodies coupled to enzymes or gold balls.
In routine human kidney biopsies, optical microscopy and immunohistochemistry (usually immunofluorescence) in the great majority of cases, allow a precise and sufficient diagnosis by a total reading of the samples.
Nevertheless, MET is still widely used in specific cases: certain hereditary diseases, diseases of “fine membranes”, dysproteinemias with organized deposits.
In practice, if the samples are sufficient, if the means are available, it is desirable, especially if there is a precise orientation, to provide for a subsequent MET study. Proper attachment and inclusion of certain specimens can then be used to keep material available for years.
Morphometry:
Morphometry is a method of measurement based on images, histological structures. For example, for the kidney, this may be a measure of the size and surface area of the glomeruli or of certain areas such as mesangial areas. The information is programmed and collected on an image analyzer, from fixed kidney sections. Quantification was performed for a large number of glomerular pathologies.
This is the case for chronic renal failure, diabetes, and toxemia. In the latter case, the measurement of the glomeruli of the toxemic women made it possible to distinguish a group of women with “small glomeruli”. This group of women will prove to be among the toxemic to a group of women who will become chronic hypertensive, several years after these measures.
Confocal microscopy:
Confocal microscopy is a technique of high precision. From very fine optical sections of cells or tissue, it can show colocalization of images corresponding to markings using different fluorochromes and interpreted in three dimensions.The simultaneous overlapping of different markings, when colocalizations exist, results in “color” changes. This technique has been used in research from renal biopsies, for example to show the transdifferentiation of podocytes in the development of certain lesions such as the HSF lesion, particularly in its cellular form, and during recurrence on the renal graft . The transdifferentiation of the podocyte corresponds to the loss of the fetal or mature phenotype of this cell and the acquisition of another phenotype. During this lesion, the podocyte acquires an “activated” macrophage phenotype (CD68 + , CD16 + , HLA + ), to which is added the epithelial cell epitope coexpression such as cytokeratins.
Molecular biology:
In situ hybridization and polymerase chain reaction (PCR) (on frozen material or paraffin according to the probes):
These techniques use complementary hybridization probes represented by complementary nucleotide sequences specific for the desired deoxyribonucleic acid (DNA) or RNA fragment. Over the past ten years, at the frontier of routine and research, they have revealed the precise place of synthesis of the protein and not its place of storage (with enzymatic revelation by labeled antibody). This technique has been used in the normal and pathological kidney to reveal the cellular synthesis of renin, conversion enzyme, urokinase, erythropoietin, tumor necrosis factor (TNF) -a, interleukin-2 and thrombin. In routine use, these probes are used for fluid and / or tissue diagnosis for a large number of viruses such as cytomegalovirus, HIV, HCV and renal transplant for BK virus. PCR and reverse transcriptase (RT) -PCR can provide positive responses for infinitesimal amounts of messenger RNA from isolated biopsy or glomerulate crusts.
“Microarrays”
Finally, great hopes are based on the latest molecular biology tools that have already proven themselves in cancer.They use techniques of microarrays (microarrays) in organ transplantation and in particular renal. They offer the possibility of characterization of a human transcriptome of the renal graft from the biopsies. In parallel with the study of immunological risk, the idea is to recognize certain genetic characteristics of the donor that can influence the immune response of the recipient. Two recent works have given rise to answers. A pediatric study has allowed to classify rejections of different gravity according to a genetic variation of certain molecules or enzymes in the graft. Another study, carried out on the basis of systematic biopsies carried out 6 months after transplantation, identified ten genes predisposing to the development of chronic allograft nephropathy.